Covalent Bond
Content:
Definition
A covalent bond meaning comes from two Latin words: “co” – jointly and “vales” – valid. Since a covalent bond occurs when a pair of electrons belonging for two atoms simultaneously. The formation of a covalent bond occurs only among the atoms of non-metals, and it can appear both in atoms of molecules and in crystals.
What Happens in a Bond?
For the first time, a covalent chemical bond was discovered back in 1916 by the American chemist J. Lewis. What did chemists find out about it? During the chemical interaction of two atoms, the transfer of electrons from one atom to another may not be possible because electronegativity of nonmetals is quite big. At this point, covalent bonds occur between the electrons of both atoms.
Types
There are two types of covalent bond:
- exchangeable,
- donor-acceptance.
The exchange type of covalent bond occurs when each of the connecting atoms represents one unpaired electron for the formation of an electron bond. In this case, these electrons must have opposite charges (spins).
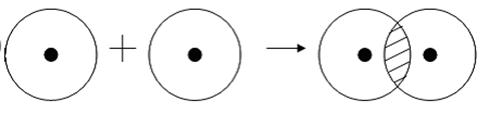
An example of such a covalent bond can be the bonds occurring in a hydrogen molecule. When hydrogen’s atoms approach each other, they penetrate into each other’s electron clouds. In chemistry, this is called overlapping electron clouds. As a result, the electron density between the nuclei increases, they themselves attract each other, and the energy of the system decreases. However, when the distance between nuclei is too close, they start to repel each other, and thus some optimal distance between them occurs.
More clearly this is shown in the picture.
The donor-acceptor type of a covalent bond occurs when one particle (the donor) represents its electron pair for the bond, and the second (the acceptor) represents a free orbital.
Nonpolar Bond
A nonpolar covalent bond definition is simple; it is a bond that is formed between two identical atoms.
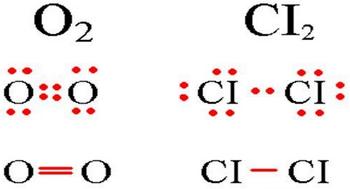
A nonpolar covalent bond picture.
In molecules with a covalent non-polar bond common electron pairs are located at equal distances from the atomic nuclei. For example, in an oxygen molecule, atoms get an eight electron configuration, and they have four common pairs of electrons.
Nonpolar covalent bond examples: gases, liquids, or relatively low melting solids.
Polar Bond
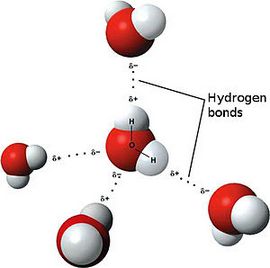
A polar covalent bond is formed when covalently bound atoms have different electronegativity, and public electrons don’t belong equally for two atoms. Most of the time, public electrons are closer to one atom than another. A polar covalent bond example: hydrogen chloride molecule, where the public electrons are located closer to the chlorine atom than hydrogen.
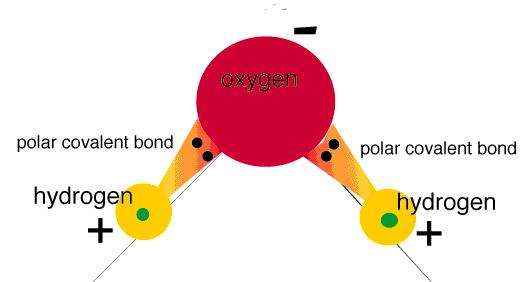
This is the polar covalent bond scheme.
Water is a prime example of a substance with a polar covalent bond.
How to Define a Bond
Well, now you know the answer to the question of how to define a polar covalent bond, and how to define a nonpolar. It’s enough to know the properties and chemical formula of molecules, if molecule consists of atoms of different elements, then the bond will be polar. If the molecule consists of one element, then the bond will be nonpolar. Besides this, it is important to remember that covalent bonds can only occur among non-metals, this is due to the mechanism of covalent bonds described above.
References and Further Reading
- Campbell, Neil A.; Williamson, Brad; Heyden, Robin J. (2006). Biology: Exploring Life. Boston, MA: Pearson Prentice Hall. ISBN 0-13-250882-6. Retrieved 2012-02-05.[better source needed]
- March, Jerry (1992). Advanced Organic Chemistry: Reactions, Mechanisms, and Structure. John Wiley & Sons. ISBN 0-471-60180-2.
- Gary L. Miessler; Donald Arthur Tarr (2004). Inorganic Chemistry. Prentice Hall. ISBN 0-13-035471-6.
- Merriam-Webster – Collegiate Dictionary (2000).
- “Chemical Bonds”. Hyperphysics.phy-astr.gsu.edu. Retrieved 2013-06-09.

Author: Pavlo Chaika, Editor-in-Chief of the journal Poznavayka
When writing this article, I tried to make it as interesting and useful as possible. I would be grateful for any feedback and constructive criticism in the form of comments to the article. You can also write your wish/question/suggestion to my mail pavelchaika1983@gmail.com or to Facebook.


Excellent job